Our universe is made of matter and energy. States of matter refers to various distinct forms in which matter can exist. Since long humans have known three basic states of matter, namely solid, liquid and gas. In 1920s, Irving Langmuir, a Nobel Prize winner in Chemistry, described fourth state of matter known as plasma. Around the same time, in 1925, Albert Einstein, crediting a pioneering paper by Indian mathematician Satyendra Nath Bose, predicted fifth state of matter which we now refer to as Bose-Einstein Condensate (BEC).
Fundamental States of Matter
Solid, liquid and gas are the states in which matter is present all around us. Matter does not need special conditions to exists in these states. We can say that solid, liquid and gas are the three common or basic states of matter. Add plasma as well to this list. Plasma is fourth fundamental state of the matter.
1. Solid
Particles in solid state are tightly bound with each other so strongly that they do not move at all. Solid state particles can only vibrate. This lack of movement gives solids their definite shape and definite volume. Solid state matter changes shape only because of external cause. Examples of solids are iron, and wood.

State of Matter: Solid
2. Liquid
Liquid particles are relatively freer and they can move a little bit. Liquid matter is incompressible fluid. When a solid is heated above its melting point, it becomes liquid. Unlike solid matter, liquids do not have a definite shape rather their shape is determined by the shape of the container. If we heat a liquid at constant pressure to the boiling point, it will convert into gas state. Examples of liquid at room temperature are water and mercury.
State of Matter: Liquid
3. Gas
Matter in gas state is a compressible fluid. Gases do not have a definite shape or volume. Particles in gas are well separated and have no regular arrangement. Gas particles can move around freely at significant speed. Like liquids, the gas matter will also take the shape of the container; but unlike liquids, the gas matter expands to fill the whole volume of the container. Examples of gases are hydrogen and nitrogen.

State of Matter: Gas
4. Plasma
Plasma is the fourth fundamental state of the matter. Matter in plasma does not have definite shape or volume. In plasma, positively charged nuclei floats in a sea of free-moving, disassociated electrons. As a result, plasma produces electric current and magnetic field. Gas can be converted into plasma by application of huge voltage difference or extreme temperature. Although plasma does not exist under normal conditions on Earth, it is commonly produced in lightening, florescent lights and plasma TV. Sun’s corona also has plasma. Plasma could in fact be the most common state of matter in the whole universe.
Fifth State of Matter
Bose-Einstein condensate (BEC) is often referred to as the fifth state of matter. When matter is cooled very nearly to absolute zero temperature −273.15 °C (−459.67 °F), the molecular movements come to a halt and all the atoms collapse to create a single “super atom”. The first BEC was created in 1995, by the research groups of Eric Cornell and Carl Wieman, of JILA at the University of Colorado at Boulder. They cooled rubidium to absolute zero to make the BEC. The Bose-Einstein condensate is used for studying quantum mechanics.
Changing State or Phase of Matter
Matter can phase from one state to another depending on the surrounding circumstances. For example, solid can convert into liquid above its melting point. Similarly liquid can convert into gas above its boiling point. Very high temperature or voltage can convert gas into plasma and extremely low temperature can create BEC. Pressure also plays a very important role in keeping or changing the state of matter.
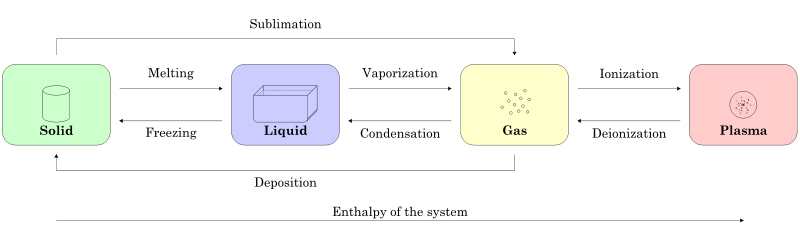
More Exotic States of Matter
Matter can exists in a large number of states. Some of these are superfluid, dropleton, glass, liquid crystal, Fermionic condensate, Rydberg molecule, quantum Hall state and photonic matter just to name a few.
Use the citation below to add this article to your bibliography
"States of Matter: Solid, Liquid, Gas, Plasma, BEC and More!." Dashamlav.com. Web. 27 July 2024. <https://dashamlav.com/matter-states-solid-liquid-gas-plasma-bec/>
Dashamlav.com, "States of Matter: Solid, Liquid, Gas, Plasma, BEC and More!." Accessed 27 July 2024. https://dashamlav.com/matter-states-solid-liquid-gas-plasma-bec/
"States of Matter: Solid, Liquid, Gas, Plasma, BEC and More!." (n.d.). Dashamlav.com. Retrieved 27 July 2024 from https://dashamlav.com/matter-states-solid-liquid-gas-plasma-bec/
