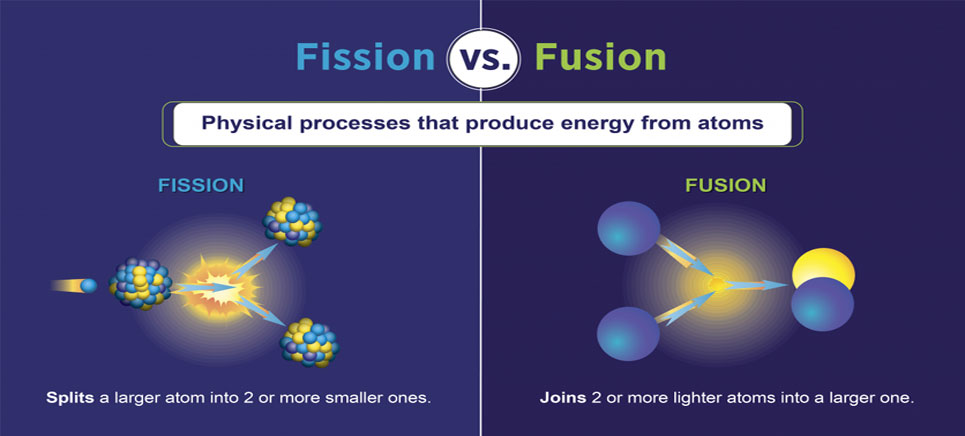
Once upon a time it was believed that atom is the smallest entity and it can not be broken any further. However by observing nuclear fission, we learnt that even atom can be broken into smaller atoms. On the other hand, atoms also combine, by a process called nuclear fusion, to create heavier elements. The following table demonstrate the major differences between these two processes.
| Base of Comparison | Nuclear Fusion | Nuclear Fission |
|---|---|---|
| Definition | Fusion is the process of combining two or more lighter atoms to create bigger heavier atoms. | Fission is the process of splitting an atom into lighter atoms. |
| Example | Nuclear fusion occurs in the core of our star — the Sun. | Nuclear fission occurs in the present day nuclear reactors. |
| Byproducts | Fusion does not create many radioactive elements as byproduct. | Fission creates many highly radioactive elements as byproduct. |
| Energy Required | Fusion needs huge amount of energy to kick-start. | Fission needs relatively much less energy to kick-start the process. |
| Energy Released | Energy released during the nuclear fusion is comparatively much higher than the fission process. | Energy released in fission is much lesser than in case of fusion. |
| Conditions Required | Fusion requires very high density, very high temperature and suitable light-weight atoms. | Fission requires atoms with critical mass and neutrons with high speed. |
| Natural Occurrence | Nuclear fusion occurs in all stars. | Nuclear fission does not occur naturally. |
| Fuel | Hydrogen isotopes (Deuterium and Tritium) are used in experimental reactions. | Uranium is mostly used in nuclear reactors to produce electricity. |
Diagram of Nuclear Fusion and Fission
Use the citation below to add this article to your bibliography
"Nuclear Fusion and Fission: Differences with Examples and Diagrams." Dashamlav.com. Web. 27 July 2024. <https://dashamlav.com/fusion-vs-fission-differences-table-examples-diagram/>
Dashamlav.com, "Nuclear Fusion and Fission: Differences with Examples and Diagrams." Accessed 27 July 2024. https://dashamlav.com/fusion-vs-fission-differences-table-examples-diagram/
"Nuclear Fusion and Fission: Differences with Examples and Diagrams." (n.d.). Dashamlav.com. Retrieved 27 July 2024 from https://dashamlav.com/fusion-vs-fission-differences-table-examples-diagram/
